Search
- Page Path
- HOME > Search
Original Articles
- Clinical Study
- Romosozumab in Postmenopausal Korean Women with Osteoporosis: A Randomized, Double-Blind, Placebo-Controlled Efficacy and Safety Study
- Ki-Hyun Baek, Yoon-Sok Chung, Jung-Min Koh, In Joo Kim, Kyoung Min Kim, Yong-Ki Min, Ki Deok Park, Rajani Dinavahi, Judy Maddox, Wenjing Yang, Sooa Kim, Sang Jin Lee, Hyungjin Cho, Sung-Kil Lim
- Endocrinol Metab. 2021;36(1):60-69. Published online February 24, 2021
- DOI: https://doi.org/10.3803/EnM.2020.848
- 6,876 View
- 390 Download
- 7 Web of Science
- 10 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
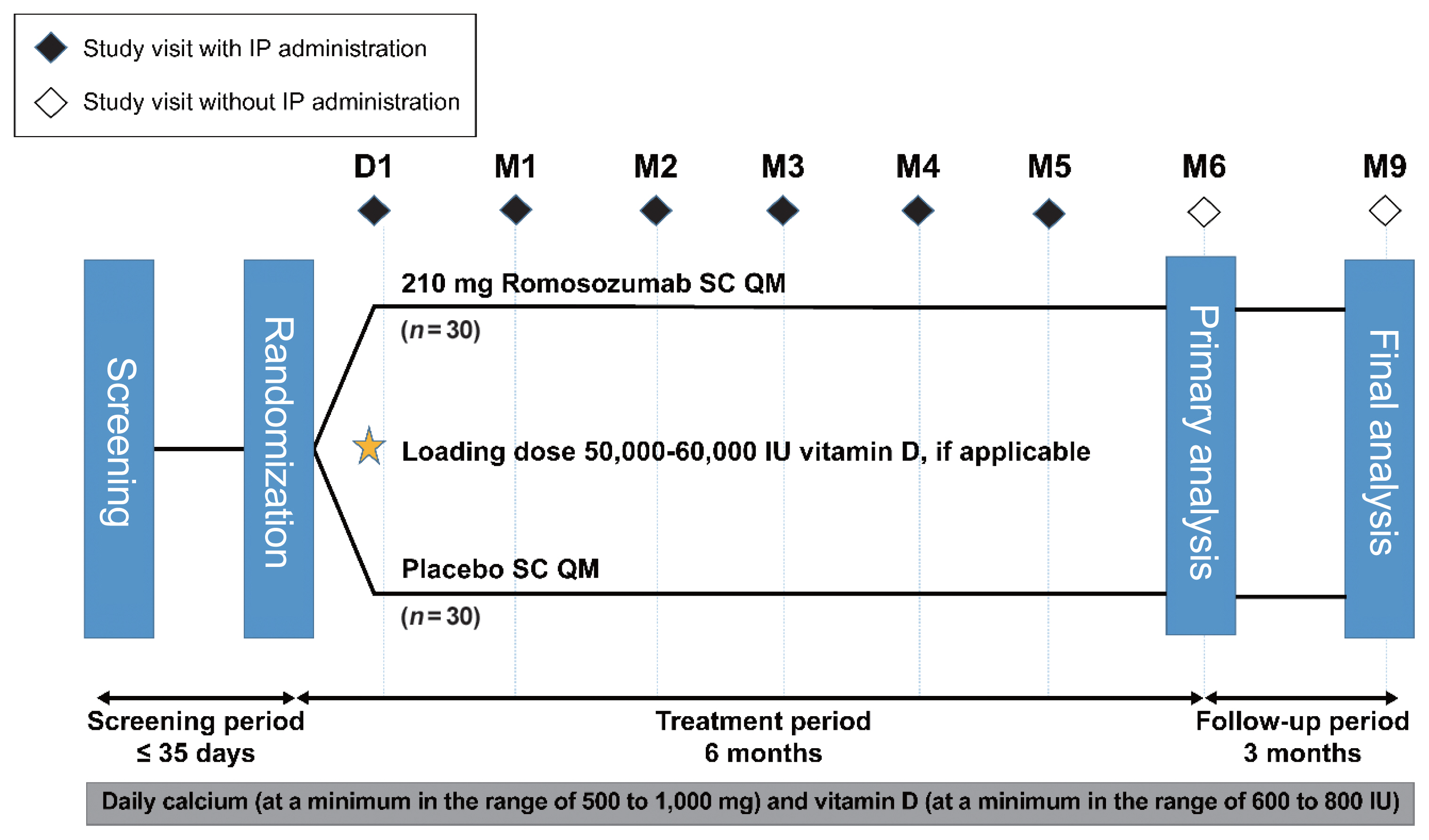
This phase 3 study evaluated the efficacy and safety of 6-month treatment with romosozumab in Korean postmenopausal women with osteoporosis.
Methods
Sixty-seven postmenopausal women with osteoporosis (bone mineral density [BMD] T-scores ≤–2.5 at the lumbar spine, total hip, or femoral neck) were randomized (1:1) to receive monthly subcutaneous injections of romosozumab (210 mg; n=34) or placebo (n=33) for 6 months.
Results
At month 6, the difference in the least square (LS) mean percent change from baseline in lumbar spine BMD (primary efficacy endpoint) between the romosozumab (9.5%) and placebo (–0.1%) groups was significant (9.6%; 95% confidence interval, 7.6 to 11.5; P<0.001). The difference in the LS mean percent change from baseline was also significant for total hip and femoral neck BMD (secondary efficacy endpoints). After treatment with romosozumab, the percent change from baseline in procollagen type 1 N-terminal propeptide transiently increased at months 1 and 3, while that in C-terminal telopeptide of type 1 collagen showed a sustained decrease. No events of cancer, hypocalcemia, injection site reaction, positively adjudicated atypical femoral fracture or osteonecrosis of the jaw, or positively adjudicated serious cardiovascular adverse events were observed. At month 9, 17.6% and 2.9% of patients in the romosozumab group developed binding and neutralizing antibodies, respectively.
Conclusion
Treatment with romosozumab for 6 months was well tolerated and significantly increased lumbar spine, total hip, and femoral neck BMD compared with placebo in Korean postmenopausal women with osteoporosis (ClinicalTrials.gov identifier NCT02791516). -
Citations
Citations to this article as recorded by- A pharmacovigilance analysis of FDA adverse event reporting system events for romosozumab
Zepeng Chen, Ming Li, Shuzhen Li, Yuxi Li, Junyan Wu, Kaifeng Qiu, Xiaoxia Yu, Lin Huang, Guanghui Chen
Expert Opinion on Drug Safety.2023; 22(4): 339. CrossRef - Evaluation of the efficacy and safety of romosozumab (evenity) for the treatment of osteoporotic vertebral compression fracture in postmenopausal women: A systematic review and meta‐analysis of randomized controlled trials (CDM‐J)
Wenbo Huang, Masashi Nagao, Naohiro Yonemoto, Sen Guo, Takeshi Tanigawa, Yuji Nishizaki
Pharmacoepidemiology and Drug Safety.2023; 32(6): 671. CrossRef - Efficacy and Cardiovascular Safety of Romosozumab: A Meta-analysis and Systematic Review
Seo-Yong Choi, Jeong-Min Kim, Sang-Hyeon Oh, Seunghyun Cheon, Jee-Eun Chung
Korean Journal of Clinical Pharmacy.2023; 33(2): 128. CrossRef - Clinical Studies On Romosozumab: An Alternative For Individuals With A High Risk Of Osteoporotic Fractures: A Current Concepts Review (Part I)
E. Carlos Rodriguez-Merchan, Alonso Moreno-Garcia, Hortensia De la Corte-Rodriguez
SurgiColl.2023;[Epub] CrossRef - Romosozumab in osteoporosis: yesterday, today and tomorrow
Dong Wu, Lei Li, Zhun Wen, Guangbin Wang
Journal of Translational Medicine.2023;[Epub] CrossRef - Efficacy and safety of anti-sclerostin antibodies in the treatment of osteoporosis: A meta-analysis and systematic review
Frideriki Poutoglidou, Efthimios Samoladas, Nikolaos Raikos, Dimitrios Kouvelas
Journal of Clinical Densitometry.2022; 25(3): 401. CrossRef - Benefits of lumican on human bone health: clinical evidence using bone marrow aspirates
Yun Sun Lee, So Jeong Park, Jin Young Lee, Eunah Choi, Beom-Jun Kim
The Korean Journal of Internal Medicine.2022; 37(4): 821. CrossRef - What is the risk of cardiovascular events in osteoporotic patients treated with romosozumab?
I. R. Reid
Expert Opinion on Drug Safety.2022; 21(12): 1441. CrossRef - Proxied Therapeutic Inhibition on Wnt Signaling Antagonists and Risk of Cardiovascular Diseases: Multi-Omics Analyses
Yu Qian, Cheng-Da Yuan, Saber Khederzadeh, Ming-Yu Han, Hai-Xia Liu, Mo-Chang Qiu, Jian-Hua Gao, Wei-Lin Wang, Yun-Piao Hou, Guo-Bo Chen, Ke-Qi Liu, Lin Xu, David Karasik, Shu-Yang Xie, Hou-Feng Zheng
SSRN Electronic Journal .2022;[Epub] CrossRef - Multi-Omics Analyses Identify Pleiotropy and Causality Between Circulating Sclerostin and Atrial Fibrillation
Yu Qian, Peng-Lin Guan, Saber Khederzadeh, Ke-Qi Liu, Cheng-Da Yuan, Ming-Yu Han, Hai-Xia Liu, Mo-Chang Qiu, Jian-Hua Gao, Wei-Lin Wang, Yun-Piao Hou, Guo-Bo Chen, Lin Xu, David Karasik, Shu-Yang Xie, sheng zhifeng, Hou-Feng Zheng
SSRN Electronic Journal .2022;[Epub] CrossRef
- A pharmacovigilance analysis of FDA adverse event reporting system events for romosozumab

- Clinical Study
- Low Predictive Value of FRAX Adjusted by Trabecular Bone Score for Osteoporotic Fractures in Korean Women: A Community-Based Cohort Study
- Hana Kim, Jung Hee Kim, Min Joo Kim, A Ram Hong, HyungJin Choi, EuJeong Ku, Ji Hyun Lee, Chan Soo Shin, Nam H. Cho
- Endocrinol Metab. 2020;35(2):359-366. Published online June 24, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.2.359
- 5,988 View
- 132 Download
- 9 Web of Science
- 10 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
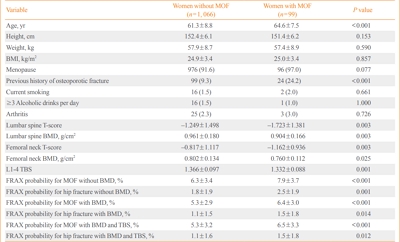
The value of the Fracture Risk Assessment Tool (FRAX) and the trabecular bone score (TBS) for assessing osteoporotic fracture risk has not been fully elucidated in Koreans. We conducted this study to clarify the predictive value of FRAX adjusted by TBS for osteoporotic fractures in Korean women.
Methods
After screening 7,192 eligible subjects from the Ansung cohort, 1,165 women aged 45 to 76 years with available bone mineral density (BMD) and TBS data were enrolled in this study. We assessed their clinical risk factors for osteoporotic fractures and evaluated the predictive value of FRAX with or without BMD and TBS.
Results
During the mean follow-up period of 7.5 years, 99 (8.5%) women suffered major osteoporotic fractures (MOFs) and 28 (2.4%) experienced hip fractures. FRAX without BMD, BMD-adjusted FRAX, and TBS-adjusted FRAX were significantly associated with the risk of MOFs (hazard ratio [HR] per percent increase, 1.08; 95% confidence interval [CI], 1.03 to 1.14; HR, 1.09; 95% CI, 1.03 to 1.15; and HR, 1.07; 95% CI, 1.02 to 1.13, respectively). However, BMD-adjusted FRAX and TBS-adjusted FRAX did not predict MOFs better than FRAX without BMD based on the Harrell’s C statistic. FRAX probabilities showed limited value for predicting hip fractures. The cut-off values of FRAX without BMD, FRAX with BMD, and FRAX with BMD adjusted by TBS for predicting MOFs were 7.2%, 5.0%, and 6.7%, respectively.
Conclusion
FRAX with BMD and TBS adjustment did not show better predictive value for osteoporotic fractures in this study than FRAX without adjustment. Moreover, the cut-off values of FRAX probabilities for treatment might be lower in Korean women than in other countries. -
Citations
Citations to this article as recorded by- Update on the utility of trabecular bone score (TBS) in clinical practice for the management of osteoporosis: a systematic review by the Egyptian Academy of Bone and Muscle Health
Yasser El Miedany, Walaa Elwakil, Mohammed Hassan Abu-Zaid, Safaa Mahran
Egyptian Rheumatology and Rehabilitation.2024;[Epub] CrossRef - Comparison of predictive value of FRAX, trabecular bone score, and bone mineral density for vertebral fractures in systemic sclerosis: A cross-sectional study
Kyung-Ann Lee, Hyun-Joo Kim, Hyun-Sook Kim
Medicine.2023; 102(2): e32580. CrossRef - Screening for the primary prevention of fragility fractures among adults aged 40 years and older in primary care: systematic reviews of the effects and acceptability of screening and treatment, and the accuracy of risk prediction tools
Michelle Gates, Jennifer Pillay, Megan Nuspl, Aireen Wingert, Ben Vandermeer, Lisa Hartling
Systematic Reviews.2023;[Epub] CrossRef - Chronic airway disease as a major risk factor for fractures in osteopenic women: Nationwide cohort study
Sung Hye Kong, Ae Jeong Jo, Chan Mi Park, Kyun Ik Park, Ji Eun Yun, Jung Hee Kim
Frontiers in Endocrinology.2023;[Epub] CrossRef - Update on the clinical use of trabecular bone score (TBS) in the management of osteoporosis: results of an expert group meeting organized by the European Society for Clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Disease
Enisa Shevroja, Jean-Yves Reginster, Olivier Lamy, Nasser Al-Daghri, Manju Chandran, Anne-Laurence Demoux-Baiada, Lynn Kohlmeier, Marie-Paule Lecart, Daniel Messina, Bruno Muzzi Camargos, Juraj Payer, Sansin Tuzun, Nicola Veronese, Cyrus Cooper, Eugene V.
Osteoporosis International.2023; 34(9): 1501. CrossRef - Comparison of HU histogram analysis and BMD for proximal femoral fragility fracture assessment: a retrospective single-center case–control study
Sun-Young Park, Hong Il Ha, Injae Lee, Hyun Kyung Lim
European Radiology.2022; 32(3): 1448. CrossRef - Association of Trabecular Bone Score-Adjusted Fracture Risk Assessment Tool with Coronary Artery Calcification in Women
Tzyy-Ling Chuang, Yuh-Feng Wang, Malcolm Koo, Mei-Hua Chuang
Diagnostics.2022; 12(1): 178. CrossRef - Risk of osteoporotic fracture in women using the FRAX tool with and without bone mineral density score in patients followed at a tertiary outpatient clinic ‒ An observational study
Maria Helena Sampaio Favarato, Maria Flora de Almeida, Arnaldo Lichtenstein, Milton de Arruda Martins, Mario Ferreira Junior
Clinics.2022; 77: 100015. CrossRef - Comparison of Trabecular Bone Score–Adjusted Fracture Risk Assessment (TBS-FRAX) and FRAX Tools for Identification of High Fracture Risk among Taiwanese Adults Aged 50 to 90 Years with or without Prediabetes and Diabetes
Tzyy-Ling Chuang, Mei-Hua Chuang, Yuh-Feng Wang, Malcolm Koo
Medicina.2022; 58(12): 1766. CrossRef - Application of the Trabecular Bone Score in Clinical Practice
Sung Hye Kong, Namki Hong, Jin-Woo Kim, Deog Yoon Kim, Jung Hee Kim
Journal of Bone Metabolism.2021; 28(2): 101. CrossRef
- Update on the utility of trabecular bone score (TBS) in clinical practice for the management of osteoporosis: a systematic review by the Egyptian Academy of Bone and Muscle Health


 KES
KES

 First
First Prev
Prev



